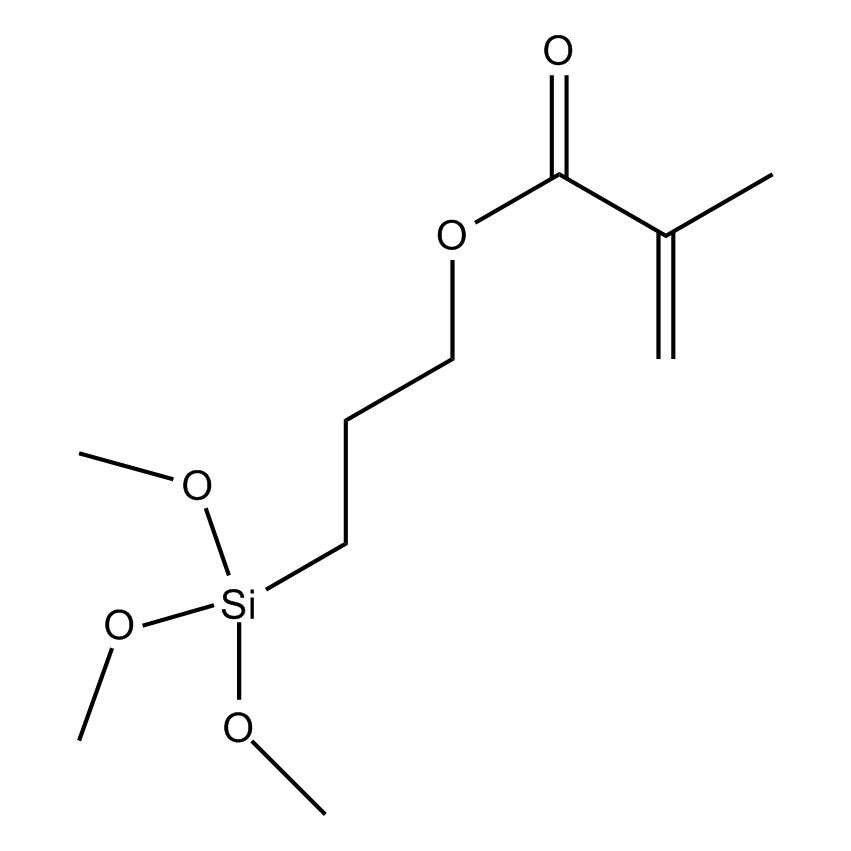
Additional Properties
- Einecs Number 219-785-8
- HMIS 3-1-1-X
- Molecular Formula C10H20O5Si
- Molecular Weight (g/mol) 248.35
- Purity (%) 97%
- TSCA Yes
- Boiling Point (˚C/mmHg) 78-81/1
- Density (g/mL) 1.045
- Flash Point (˚C) 108 °C
- Melting Point (˚C) -48°
- Refractive Index @ 20˚C 1.4310
- Specific Wetting (m2/g) 314
- Viscosity at 25 ˚C (cSt) 2
Application
Widely used coupling agent for unsaturated polyester-fiberglass composites.1
Copolymerized with styrene in formation of sol-gel composites.2
Employed in dental polymer composites.3
Reference
1. Arkles, B. Chemtech 1977, 7, 713.
2. Wei, Y. et al. J. Mater. Res. 1993, 8, 1143.
3. Matinlinna, J. et al. Int. J. Prosthodontics 2004, 17, 157.
Safety
Methacrylate Functional Trialkoxy Silane
Silane coupling agents have the ability to form a durable bond between organic and inorganic materials to generate desired heterogeneous environments or to incorporate the bulk properties of different phases into a uniform composite structure. The general formula has two classes of functionality. The hydrolyzable group forms stable condensation products with siliceous surfaces and other oxides such as those of aluminum, zirconium, tin, titanium, and nickel. The organofunctional group alters the wetting or adhesion characteristics of the substrate, utilizes the substrate to catalyze chemical transformations at the heterogeneous interface, orders the interfacial region, or modifies its partition characteristics, and significantly effects the covalent bond between organic and inorganic materials.
Methacryloxypropyltrimethoxysilane, 3-(Trimethoxysilyl)propyl methacrylate, MEMO