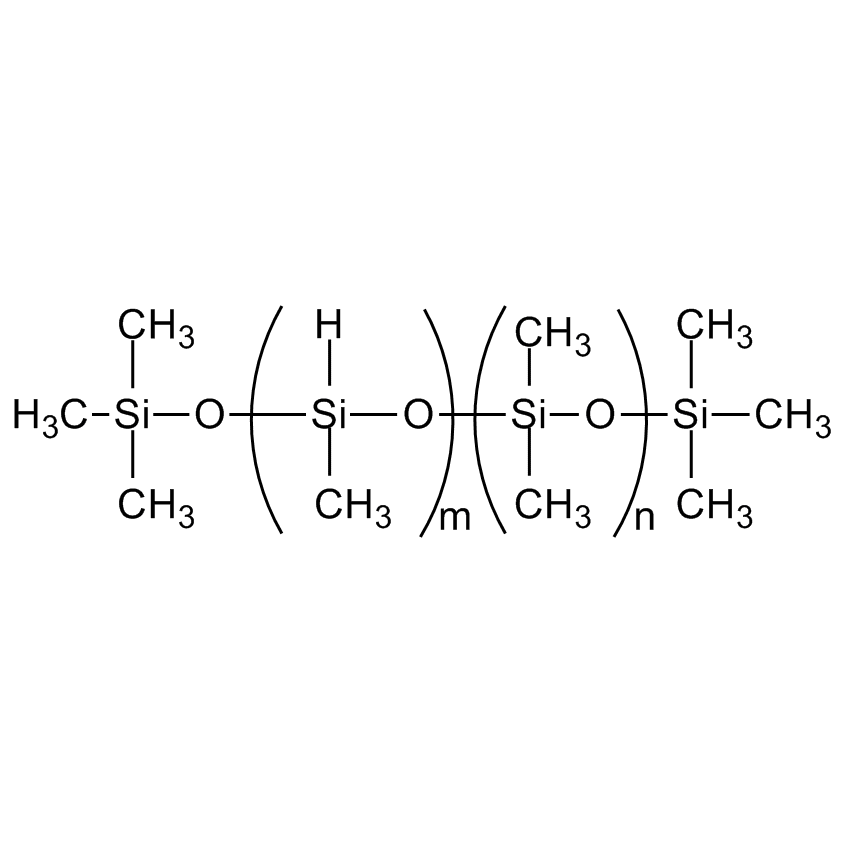Safety
(4-8% METHYLHYDROSILOXANE) - DIMETHYLSILOXANE COPOLYMER, TRIMETHYLSILOXANE TERMINATED, 6,000-9,000 cSt
- HMIS 1-1-1-X
- Molecular Weight (g/mol) 50,000-60,000
- TSCA Yes
- Boiling Point (˚C/mmHg) NA
- Density (g/mL) 0.97
- Refractive Index @ 20˚C 1.403
- Viscosity at 25 ˚C (cSt) '6,000-9,000
Siloxane-Based Silane Reducing Agent
Organosilanes are hydrocarbon-like and possess the ability to serve as both ionic and free-radical reducing agents. These reagents and their reaction by-products are safer and more easily handled and disposed than many other reducing agents. The metallic nature of silicon and its low electronegativity relative to hydrogen lead to polarization of the Si-H bond yielding a hydridic hydrogen and a milder reducing agent compared to aluminum-, boron-, and other metal-based hydrides. A summary of some key silane reductions are presented in Table 1 of the Silicon-Based Reducing Agents brochure.
Pendant Functional Fluid; MethylHydrosiloxane-Dimethylsiloxane Copolymer, TMS Terminated
MethylHydrosiloxane copolymers are the primary cross linkers for 2-part vinyl-addition silicones and intermediates for functional copolymers.
Hydrogen dimethicone; Dimethylhydropolysiloxane; Dimethicone/methicone copolymer; Polysiloxanes dimethyl methyl hydrogen, Hydride functional polydimethylsiloxane; Siloxanes and silicones, dimethyl, methyl hydrogen